Dear colleagues,
It is a pleasure to announce the publication of a new scientific paper from our colleagues of DRL - German Aerospace Agency.
It is a very important achievement for the fight against COVID and high transmissibility viruses in general. We want to congratulate with all our colleagues coordinated by Dr. Frank Duschek. We are proud about the cooperation with DLR in the aim of CBRNe activities.
Abstract:
- Significance: Fast and reliable detection of infectious SARS-CoV-2 virus loads is an important issue. Fluorescence spectroscopy is a sensitive tool to do so in clean environments. This presumes a comprehensive knowledge of fluorescence data.
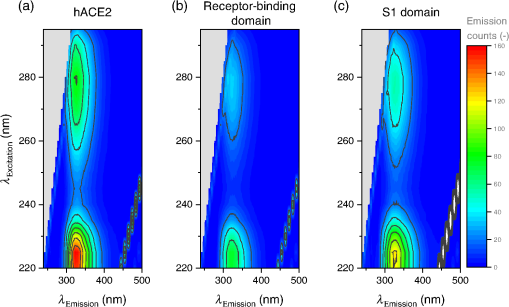
- Aim: We aim at providing fully featured information on wavelength and time-dependent data of the fluorescence of the SARS-CoV-2 spike protein S1 subunit, its receptor-binding domain (RBD), and the human angiotensin-converting enzyme 2, especially with respect to possible optical detection schemes.
- Approach: Spectrally resolved excitation-emission maps of the involved proteins and measurements of fluorescence lifetimes were recorded for excitations from 220 to 295 nm. The fluorescence decay times were extracted by using a biexponential kinetic approach. The binding process in the SARS-CoV-2 RBD was likewise examined for spectroscopic changes.
- Results: Distinct spectral features for each protein are pointed out in relevant spectra extracted from the excitation-emission maps. We also identify minor spectroscopic changes under the binding process. The decay times in the biexponential model are found to be ( 2.0 ± 0.1 ) ns and ( 8.6 ± 1.4 ) ns.
- Conclusions: Specific material data serve as an important background information for the design of optical detection and testing methods for SARS-CoV-2 loaded media.
STAY TUNED!

Write a comment